Trial vaccine to protect against mosquito-borne diseases
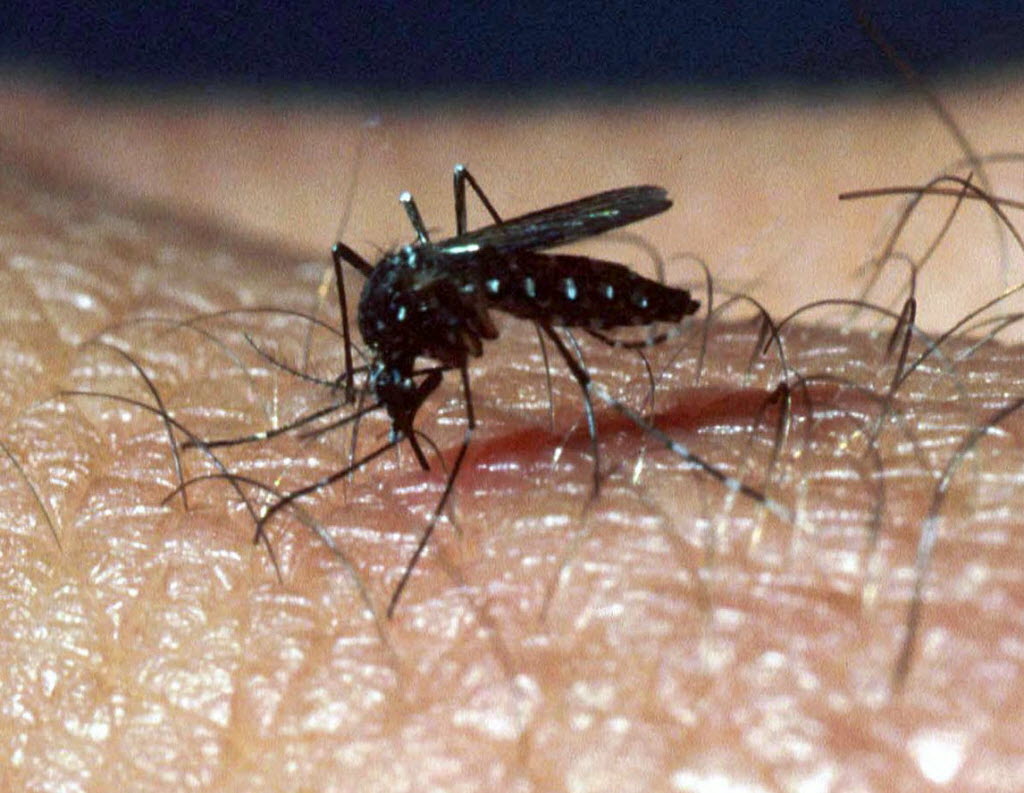
The National Institutes of Health has launched a clinical trial to test an investigational vaccine that would protect against multiple mosquito-transmitted diseases, such as Zika, malaria, West Nile and dengue fever.
The vaccine would also interfere with mosquitoes’ ability to transmit the infections.
The vaccine, called AGS-v, is designed to trigger an immune response to mosquito saliva, not the specific viruses or parasites.
“The test vaccine contains four synthetic proteins from mosquito salivary glands. The proteins are designed to induce antibodies in a vaccinated individual and to cause a modified allergic response that can prevent infection when a person is bitten by a disease-carrying mosquito,” according to the NIH.
“Mosquitoes cause more human disease and death than any other animal,” said Dr. Anthony S. Fauci, National Institute of Allergy and Infectious Diseases director, in a news release. “A single vaccine capable of protecting against the scourge of mosquito-borne diseases is a novel concept that, if proven successful, would be a monumental public health advance.”
The trial will enroll up to 60 healthy adults ages 18 to 50. Participants will receive two injections, 21 days apart. The injections will either contain the vaccine, the vaccine combined with an adjuvant (used to enhance immune responses) or sterile water.
Participants will submit blood samples to measure antibody levels between injections. After receiving the second vaccination, participants will be exposed to biting mosquitoes (The mosquitoes will not be carrying viruses or parasites.) and then submit blood samples.
Researchers will also be looking to see if the mosquitoes’ life cycle changes after exposure to the vaccine. Scientists suspect mosquitoes exposed to the vaccine may have altered behavior that could lead to early death or a reduced ability to reproduce – both of which would hinder disease transmission by controlling the mosquito population.
